# 6687
Last night the CDC posted updated laboratory test results from patients - and un-opened vials of methylprednisolone produced by the New England Compounding Center – as part of their investigation into a multi-state outbreak of fungal meningitis.
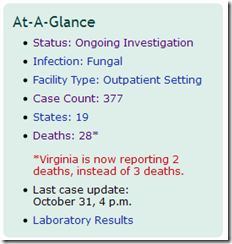
The latest numbers from this outbreak, as of 2pm yesterday, are:
Lab tests have isolated Exserohilum rostratum in 52 of the 54 patients with CDC laboratory-confirmed fungal meningitis. Tests by the CDC and FDA have also found Exserohilum rostratum in unopened vials from two of the three recalled lots of injectable steroids produced by NECC. Tests are pending on the third lot.
Here’s the complete CDC Update:
Laboratory Testing and Results from the Outbreak
October 31, 2012 6:00 PM EDT
CDC laboratory-confirmed results found on this page are from three New England Compounding Center (NECC) preservative-free methylprednisolone acetate (MPA) lots recalled on September 26, 2012.(1)
CDC scientist examines microscopic slides showing Exserohilum rostratum (on screen) during the multistate meningitis outbreak.
CDC's Fungus Reference Laboratory has the capacity and technology to examine fungal isolates under the microscope and to confirm their identification using DNA sequencing methods.
- For the multistate outbreak, the fungus laboratory is working with the Clinical and Environmental Microbiology Branch, Division of Healthcare Quality Promotion, to detect and identify other microbial pathogens. In addition, the Infectious Diseases Pathology Branch, Division of High-Consequence Pathogens and Pathology, is examining tissues from biopsies and autopsy materials.
- CDC and the states are testing tissue and fluid samples from patients with probable or confirmed fungal infection. CDC scientists are looking to see if the samples contain fungi and other microbial pathogens, and if so, what type.
- In addition, FDA and CDC are testing vials of NECC preservative-free MPA to see if they contain fungi and other microbial pathogens and if so, what type.
CDC Laboratory Results
As of October 22, 2012, Exserohilum rostratum has been found in clinical specimens for all but two of the 54 patients with CDC laboratory-confirmed fungal meningitis who meet the confirmed case definition.(2)
Tests at CDC and FDA laboratories on the preservative-free MPA vials have confirmed the presence of the same fungus, Exserohilum rostratum, in unopened vials from two of the three recalled lots.(1) Testing on the third lot of preservative-free MPA is ongoing. These laboratory test results strengthen the link between preservative-free MPA vials and the outbreak.
The fungi found in both patients and in recalled vials are common in the environment but were not recognized as a cause of meningitis before this outbreak.