# 9415
Although the CDC has warned that Early Data Suggests a Potentially Severe Flu Season ahead, and there are growing concerns of an H3N2 vaccine component `mismatch’ (see CDC HAN Advisory On `Drifted’ H3N2 Seasonal Flu Virus), North America’s flu season is just really getting started.
The latest FluView report shows modest increases in flu activity over the previous week, along with a rising percentage of `antigenically mismatched’ H3N2 virus detections (last week 52%, this week 58%).
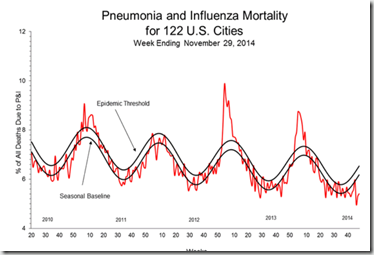
Influenza virus detections are nearly 94% Influenza A, and of those tested, they are overwhelmingly H3N2 viruses. For now, we remain well below the P&I (Pneumonia & Influenza) epidemic threshold.
Despite the possibility of seeing a less effective flu vaccine this year, the CDC continues to recommend that everyone get the shot, as it protects against more than just H3N2 subtype, and it may also provide some protection even against the drifted strain.
The MMWR reminds us, next week is National Influenza Vaccination Week — December 7–13, 2014.
As it takes a couple of weeks for a flu shot to reach its maximum level of protectiveness, it is better to get it before influenza starts circulating in your community.
And this advice on the value of antiviral treatment from yesterday’s press release from the CDC if you do get the flu:
Those at high risk from influenza include children younger than 5 years (especially those younger than 2 years); adults 65 years and older; pregnant women; and people with certain chronic health conditions such as asthma, diabetes, heart or lung disease, and kidney disease.
CDC recommends that people at high risk check with their doctor or other health care professional promptly if they get flu symptoms.
Some highlights from this week’s report include.
Weekly U.S. Influenza Surveillance Report
2014-2015 Influenza Season Week 48 ending November 29, 2014
Full report also available as PDF
Synopsis:
During week 48 (November 23-29, 2014), influenza activity increased in the United States.
- Viral Surveillance: Of 13,398 specimens tested and reported by U.S. World Health Organization (WHO) and National Respiratory and Enteric Virus Surveillance System (NREVSS) collaborating laboratories during week 48, 2,274 (17.0%) were positive for influenza.
- Pneumonia and Influenza Mortality: The proportion of deaths attributed to pneumonia and influenza (P&I) was below the epidemic threshold.
- Influenza-associated Pediatric Deaths: No influenza-associated pediatric deaths were reported.
- Influenza-associated Hospitalizations: A cumulative rate for the season of 1.8 laboratory-confirmed influenza-associated hospitalizations per 100,000 population was reported.
- Outpatient Illness Surveillance:The proportion of outpatient visits for influenza-like illness (ILI) was 2.6%, above the national baseline of 2.0%. Eight of 10 regions reported ILI at or above region-specific baseline levels. Puerto Rico and six states experienced high ILI activity; one state experienced moderate ILI activity; five states experienced low ILI activity; New York City and 38 states experienced minimal ILI activity; and the District of Columbia had insufficient data.
- Geographic Spread of Influenza: The geographic spread of influenza in Puerto Rico and six states was reported as widespread; 14 states reported regional activity; the U.S. Virgin Islands and 19 states reported local activity; the District of Columbia and 11 states reported sporadic activity; and Guam did not report.
<SNIP>
<SNIP>
A (H3N2) [114]: Forty-eight (42%) of the 114 H3N2 viruses tested have been characterized as A/Texas/50/2012-like, the influenza A (H3N2) component of the 2014-2015 Northern Hemisphere influenza vaccine. Sixty-six (58%) of the 114 viruses tested showed either reduced titers with antiserum produced against A/Texas/50/2012 or belonged to a genetic group that typically shows reduced titers to A/Texas/50/2012. Among viruses that showed reduced titers with antiserum raised against A/Texas/50/2012, most were antigenically similar to A/Switzerland/9715293/2013, the H3N2 virus selected for the 2015 Southern Hemisphere influenza vaccine. A/Switzerland/9715293/2013 is related to, but antigenically and genetically distinguishable, from the A/Texas/50/2012 vaccine virus. A/Switzerland-like H3N2 viruses were first detected in the United States in small numbers in March of 2014 and began to increase through the spring and summer.
<SNIP>
During week 48, the following ILI activity levels were experienced:
- Puerto Rico and six states (Alabama, Florida, Georgia, Louisiana, Mississippi, and Texas) experienced high ILI activity.
- One state (Illinois) experienced moderate ILI activity.
- Five states (Kansas, Missouri, Nevada, Utah, and Virginia) experienced low ILI activity.
- New York City and 38 states (Alaska, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Hawaii, Idaho, Indiana, Iowa, Kentucky, Maine, Maryland, Massachusetts, Michigan, Minnesota, Montana, Nebraska, New Hampshire, New Jersey, New Mexico, New York, North Carolina, North Dakota, Ohio, Oklahoma, Oregon, Pennsylvania, Rhode Island, South Carolina, South Dakota, Tennessee, Vermont, Washington, West Virginia, Wisconsin, and Wyoming) experienced minimal ILI activity.
- Data were insufficient to calculate an ILI activity level from the District of Columbia.