# 9813
Although well known (and respected) by infectious disease geeks, yesterday’s publication in Nature: Dissemination, Divergence & Establishment of H7N9 In China has suddenly thrust this H7 avian flu virus into the limelight, casting it in the role of the potentially driving the next influenza pandemic.
It isn’t alone, of course. There are other pandemic contenders out there, including the venerable H5N1 virus.
But H7N9’s rapid spread, and growing genetic diversity in China – combined with its ability to infect humans – has seemingly elevated it to the level of Primus inter pares – or `first among equals’ – in the pandemic flu world.
Overnight we’ve seen a plethora of headlines heralding the threat:
H7N9 bird flu has the makings of a pandemic virus, scientists warn
Scientists warn H7N9 bird flu may pose pandemic threat
While yesterday’s report in Nature provides more information, and includes a stern warning about its implications, this isn’t the first study to cast H7N9 in a possible pandemic role. Very early on after it appeared in China in the spring of 2013 we began to hear warnings from scientists, and over the past couple of years, we’ve followed the virus’s evolution.
In June of 2014, in Eurosurveillance: Genetic Tuning Of Avian H7N9 During Interspecies Transmission, we saw evidence of the genetic diversity, and continual evolution, of the H7N9 virus in Mainland China. Researchers found that at least 26 separate genotypes had emerged, mostly during the first wave, through a process they called `genetic tuning’.
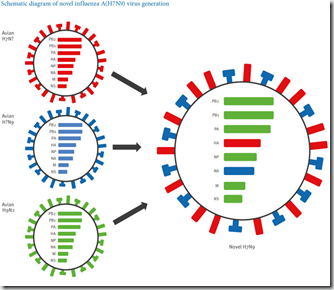
Yesterday’s report expands that array of genotypes to 48, divided among three well defined clades, and even includes a previously unrecognized subtype – H7N6 - in chickens. Previously, during the first wave in 2013, a new H7N7 virus was also recognized (see Nature: Genesis Of The H7N9 Virus).
The power, and the threat, of H7N9 isn’t simply that it could pick up the `right’ mutations and become fully transmissible in humans, thereby sparking a pandemic. It’s that H7N9 is genetically malleable enough to serve as a stepping stone – or bridge – to a completely new subtype.
As we’ve seen over the past couple of years with the sudden expansion of the HPAI H5 universe – which now encompasses H5N1, H5N2, H5N3, H5N5, H5N6, and H5N8 - H7N9 seems poised to grow the H7 flu line as well.
Two years ago we really only had one avian flu virus we worried about; H5N1. But in the spring of 2013 the novel flu field was joined by the unexpected arrival of highly pathogenic (in humans) H7N9 virus, which spread stealthily, and asymptomatically in chickens.
And over the next 12 months, we saw a parade of new subtypes begin to emerge, including H6N1, H10N8, H5N2, H5N3, H5N6 and H5N8.
The process by which these new subtypes evolve is called reassortment, and that can happen anytime a single host is simultaneously infected with two different flu subtypes.
For avian viruses, birds are the obvious host of choice, while swine flu viruses reassort primarily in pigs. But any susceptible host can produce a reassortant virus, including dogs, cats, horses, and humans.
Fortunately, most reassortant viruses are evolutionary dead ends – unable compete against existing viruses. But increasingly over the past few years, we are seeing `biologically fit’ reassortants emerge, capable of holding their own, and even thriving in the wild.
Last month, in HK’s Dr. Ko Wing-man On Flu Reassortment Concerns, we looked at the very real concerns expressed by Hong Kong’s Director of their Centre for Health Protection, that H7N9 could reassort with H3N2 during their particularly heavy flu season.
It is not an unreasonable concern. While rarely detected, co-infections with two flu subtypes do occur, and probably more often than we suspect.
Previously, in the Lancet: Coinfection With H7N9 & H3N2, we saw the first evidence of co-infection with the newly emerged H7N9 virus and a seasonal flu virus in a human. While last October, in EID Journal: Human Co-Infection with Avian and Seasonal Influenza Viruses, China, we looked at co-infections in 2 patients in Hangzhou, in January 2014.
In all of three of these cases, no reassortant virus was detected.
But In 2011, an influenza co-infection in Canada led to the creation of a unique hybrid reassorted virus (see Webinar: pH1N1 – H3N2 A Novel Influenza Reassortment), although it was not passed on to anyone else.
In recent years we’ve seen a growing list of novel (avian, swine, canine, even seal) flu viruses emerge (H5N3, H5N2, H5N5, H5N6, H5N8, H7N9, H10N8, H10N7, H3N8, H6N1, H1N1v, H1N2v, H3N2v, etc. . .), and each carries some risk of reassortment.
With other novels viruses, or with human viruses. Or conceivably both.
Over the past couple of years, the number of novel flu threats has grown dramatically, and that growth spurt shows no signs of abating. With more and more influenza subtypes, clades - and genotypes within these clades - circulating, Nature’s laboratory gets more `interchangeable parts’ to play with.
While a pandemic may not be imminent, given the amount of `viral chatter’ we are hearing, the threat level is certainly elevated. And by the time it is obvious that a pandemic threat has emerged, our `lead time’ to prepare may be down to weeks.
Two weeks ago, the World Health Organization released a statement called Warning signals from the volatile world of influenza viruses where they cautioned:
Warning: be prepared for surprises
Though the world is better prepared for the next pandemic than ever before, it remains highly vulnerable, especially to a pandemic that causes severe disease. Nothing about influenza is predictable, including where the next pandemic might emerge and which virus might be responsible. The world was fortunate that the 2009 pandemic was relatively mild, but such good fortune is no precedent.
Whether the next big global health crisis stems from H7N9, H5N1, a new flu reassortant, MERS-CoV, or `Virus X’ – the one we don’t know about . . . yet – our level of preparedness will, in large measure, determine its impact.
Now is the time for agencies, organizations, businesses, communities, and families to dust off their pandemic plans, review them, and make any needed refinements.
You do have a pandemic plan, don’t you?
For some recent pandemic preparedness blogs, you may wish to revisit:
Do You Still Have A CPO?
Pandemic Planning For Business
NPM13: Pandemic Planning Assumptions
The Pandemic Preparedness Messaging Dilemma