On Friday the ECDC published an Epidemiological Update, which focuses on this rising subvariant, and predicts it will become the dominant strain in Europe in the next 30 to 60 days. A similar trajectory seem likely in the United States and Canada as well.
First the Executive Summary, and a link to the full (7-page) PDF file. I'll have a bit more after the break.
Spread of the SARS-CoV-2 Omicron variant sub-lineage BQ.1 in the EU/EEA
Monitoring
21 Oct 2022
European Union/European Economic Area (EU/EEA) countries have detected the circulation of SARS-CoV-2 variant sub-lineages BQ.1 in levels ranging from 0−19% during week 40. This variant originates from the BA.5 Omicron Variant of Concern (VOC).
Executive summary
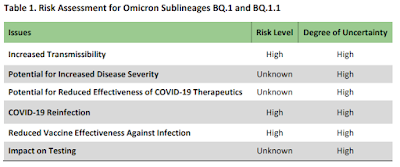
- BQ.1, including its sub-lineages, has been designated as Variant of Interest (VOI) by ECDC as of 20 October 2022. Based on modelling estimates, it is expected that by mid-November to beginning of December 2022, more than 50% of SARS-CoV-2 infections will be due to BQ.1/BQ.1.1. By the beginning of 2023, more than 80% of SARS-CoV-2 cases are expected to be due to BQ.1/BQ.1.1.
- The observed increase in the growth rate of BQ.1 is probably driven mainly by immune escape. This variant and its sub-lineages will probably contribute to a further increase in cases of COVID-19 in the EU/EEA in the coming weeks and months. The extent of the increase in COVID-19 cases will depend on various factors, including immune protection against infection influenced by the timing and coverage of COVID-19 vaccination regimes, and the extent, timing and variant landscape of previous SARS-CoV-2 pandemic waves. Based on limited available data, there is no evidence of BQ.1 being associated with a greater infection severity than the circulating variants BA.4/BA.5.
- Countries should remain vigilant for signals of BQ.1 emergence and spread; maintain sensitive and representative testing and genomic surveillance with timely sequence reporting and strengthen sentinel surveillance systems (primary care ILI/ARI and SARI).
- Countries should continue to monitor COVID-19 case rates - especially in people aged 65 years and older - and severity indicators such as hospitalisations, ICU admissions, ICU occupancy and death.
- Improving COVID-19 vaccine uptake of the primary course and first booster dose remains a priority for all eligible individuals that are not up-to-date with the recommended schedule. For the time being, for current autumn/winter vaccination campaigns, an additional booster dose should also be offered, prioritising individuals who are at risk of progression to severe disease, such as older adults (e.g. above 60 years of age), immunocompromised individuals and those with underlying medical conditions, and pregnant women. Residents and staff in long-term care facilities, as well as healthcare workers should also be considered among priority groups.
Spread of the SARS-CoV-2 Omicron variant sub-lineage BQ.1 in the EU/EEA - EN - [PDF-682.06 KB]
Since BQ.1 is an offshoot of BA.5, it is expected that the new bivalent mRNA booster shot - which contains the BA.5/BA.4 spike protein - may provide some additional protection against this emerging variant.
But how much protection, and for how long, remain unknown. `Breakthrough' infections are likely to be fairly common.
As far as severity is concerned, we haven't seen anything to suggest that BQ.1/BQ1.1 produces more serious illness than BA.5., although their prevalence in the community is still too low to know for certain.
Given what we know about `long-COVID', and the potential risks of repeated infections (see Outcomes of SARS-CoV-2 Reinfection), the severity of the acute phase of the illness isn't the only consideration.There is also evidence suggesting that BQ.1/BQ.1.1 are likely to evade our dwindling monoclonal antibody armamentarium against COVID, which may limit treatment options. This from the NIH.
The COVID-19 Treatment Guidelines Panel’s Statement on Omicron Subvariants and Anti-SARS-CoV-2 Monoclonal Antibodies
Last Updated: October 19, 2022
On October 14, 2022, the Centers for Disease Control and Prevention (CDC) reported a rapid increase in certain SARS-CoV-2 Omicron subvariants circulating in the United States1 that are likely to be resistant to some anti-SARS-CoV-2 monoclonal antibodies (mAbs). The subvariants BQ.1 and BQ.1.1 are likely to be resistant to bebtelovimab, and the subvariants BA.4.6, BA.2.75.2, BF.7, BQ.1, and BQ.1.1 are likely to be resistant to tixagevimab plus cilgavimab (Evusheld). The anticipated loss of susceptibility is based on knowledge about amino acid mutations that confer antibody resistance and on available data from in vitro neutralization studies.2
While BQ.1/BQ.1.1 appears headed for dominance in the short run, COVID variants in the age of Omicron have all enjoyed relatively short reigns. BA.5 has hung on longer than most, but on average, we've seen a new rising star emerge every 3 or 4 months.
As long as this rapid-replacement cycle continues, any talk of an end to the COVID pandemic is likely premature.