#14,557
In March of 2013, in EID Journal: Predicting Hotspots for Influenza Virus Reassortment, both China and Egypt were at the top of the list of areas likely to spawn the next novel flu virus.
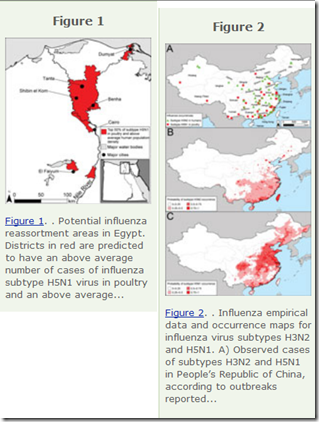 | ||||||||
| EID Influenza Hotspots For Reassortment - 2013 |
While China has since gotten most of our attention, rapidly producing H10N8, H5N6, and H5N8 (to name a few), between November 2014 and May 2015, Egypt saw the biggest outbreak of human infection with H5N1 on record (see EID Dispatch: Increased Number Of Human H5N1 Infection – Egypt, 2014-15).
Both countries lie beneath major migratory flyways, both are major poultry producers (with many backyard flocks), and both have numerous LPAI and HPAI subtypes circulating in local birds.While China appears to have gained better control of avian flu the past few years, Egypt continues to struggle with spotty poultry vaccination campaigns that often rely on older, mismatched vaccines (see Egypt: A Paltry Poultry Vaccine), which can potentially drive vaccine escape mutations.
As a result, multiple clades of HPAI H5 viruses (H5N1 and H5N8) - along with H9N2 - have all become endemic in Egyptian poultry.In early March of this year, in Egyptian MOA: Reassorted H5N2 Detected On Duck Farm, we saw a short statement announcing the detection of a new H5N2 virus on an Egyptian duck farm, followed 3 days later by a barebones OIE Notification.
That announcement followed similar reports from Russia, which in December of 2017 and again in August 2018, reported outbreaks of HPAI H5N2 in poultry farms in Kostroma Oblast.The OIE confirmed that the second Russian H5N2 virus was of clade 2.3.4.4. - which suggested it was a relatively new reassortment - likely from either HPAI H5N8 or HPAI H5N6.
In June (see Viruses: A Novel Reassortant H5N2 Virus In Egypt), we got our first detailed look at the Egypt's new H5N2 virus (A/duck/Egypt/VG1099/2018), published in the journal Viruses. As with the Russian H5N2, it was of clade 2.3.4.4.
More specifically, a reassortant between HPAI H5N8 and LPAI H9N2, both of which - along with HPAI H5N1 - co-circulate in Egypt.Today we've a new study, which describes an entirely different reassorted H5N8/H9N2 virus, in broiler farms in Egypt. A concern because up until earlier this year, HPAI H5 and H9N2 have appeared to co-circulate in Egypt without reassorting.
A link, and some excerpts from today's EID dispatch, then I'll return with a postscript.
Volume 26, Number 1—January 2020
Dispatch
Novel Reassortant Highly Pathogenic Avian Influenza A(H5N2) Virus in Broiler Chickens, Egypt
Kareem E. Hassan, Jacqueline King, Magdy El-Kady, Manal Afifi, Hassanein H. Abozeid, Anne Pohlmann, Martin Beer, and Timm Harder
Abstract
We detected a novel reassortant highly pathogenic avian influenza A(H5N2) virus in 3 poultry farms in Egypt. The virus carried genome segments of a pigeon H9N2 influenza virus detected in 2014, a nucleoprotein segment of contemporary chicken H9N2 viruses from Egypt, and hemagglutinin derived from the 2.3.4.4b H5N8 virus clade.
Since 2006, Egypt’s poultry industry has been plagued by endemic infections with highly pathogenic avian influenza (HPAI) virus, subtype H5N1, clade 2.2.1, of the goose/Guangdong (gs/GD) lineage (1). In addition, low pathogenicity avian influenza (LPAI) virus of subtype H9N2, G1 lineage, introduced in 2011 (2), and HPAI H5N8 (gs/GD clade 2.3.4.4b) introduced in 2016, have become entrenched in local poultry populations (3). Despite ongoing control measures, respiratory disease with increased mortality rates is endemic in poultry farms in Egypt.
The zoonotic nature of HPAI H5N1 2.2.1 viruses has caused in Egypt the highest number of human infection cases per country worldwide; a low level of sporadic benign human cases of H9N2 viral infection has also been reported from Egypt (4). Continued adaptation by point mutations, but not reassortment, to enhance replication in mammalian hosts has been repeatedly reported in avian influenza in Egypt (5). Here, we describe the detection of a new reassortant HPAI virus in commercial chicken holdings in Egypt. This virus carries the hemagglutinin (HA) gene of HPAI clade 2.3.4.4b H5N8 virus and 7 genome segments derived from Egyptian H9N2 viruses (6).
(SNIP)
In March 2019, Egypt’s Ministry of Agriculture announced the detection of a new influenza A(H5N2) virus from seemingly healthy ducks in the Dakahlia governorate (11); recently published information on this reassortant indicated the presence of a neuraminidase N2 segment of chicken H9N2 viruses in the background of an HPAI H5 clade 2.3.4.4b virus (11).
Our data confirm the presence of a different H5N2 reassortant and its occurrence in chickens in different geographic regions of Egypt (Table). We detected the current reassortant HPAI H5N2 viruses in 2 different broiler farms in Beheira (January and March 2019) and 1 broiler farm in Fayoum (January 2019) governorates (Table).
The HA amino acid sequence of these reassortants does not signal antigenic variation compared with parent HPAI H5 subtype of clade 2.3.4.4b. Antigenic and further phenotypic properties, such as host specificity, require investigation as soon as isolates are available. For the H5N2-positive samples, only FTA card material was available at Friedrich-Loeffler-Institut. However, H5N2 isolates were successfully generated at the Beni-Suef University, Egypt, but were currently not available for further antigenic and phenotypic analyses (M. El-Kady, unpub. data).
Intensified targeted surveillance in poultry and pigeons is urgently required and may lead to detection of additional reassortants. However, co-detection in a sample of H5N8 and H9N2 subtypes by reverse transcription quantitative PCR may blur the identification of H5N2 reassortants; plaque purification of such samples would aid in separating subtypes but cannot currently be used in routine diagnostics.
There is a risk for transboundary spread of HPAI A(H5N2) virus in northern Africa and the Middle East, and similar reassortment events are to be expected in regions where clade 2.3.4.4 HPAI and H9N2 viruses are co-circulating.
Long-term solutions in combating avian influenza virus infections in poultry are sorely needed and would help to lower risks of human exposure to zoonotic avian influenza viruses such as the highly zoonotic H7N9 viruses in China that carry a full set of internal genes of an H9N2-G1–like avian influenza virus (12).
Dr. Hassan is a poultry veterinary specialist at Beni-Suef University, currently on leave to complete his PhD studies at the Friedrich-Loeffler-Institut, Isle of Riems, Germany. His primary research interest is focused on respiratory infectious diseases in poultry with an emphasis on avian influenza.
LPAI H9N2 is ubiquitous in Asia and across much of the Middle East, and while it currently is viewed as being a relatively minor pandemic threat (see CDC IRAT SCORE), over the past few years, we've seen it acquire new, and worrying mammalian adaptations.
PLoS Path.: PB2 Mutations in H9N2 Enhance Replication & Growth In Mammals
Virology Journal: Mouse-adapted H9N2 Avian Influenza Virus Causes Systemic Infection in Mice
Viruses: Characterization of the H9N2 Avian Influenza Viruses Currently Circulating in South ChinaOf the HPAI viruses of greatest concern over the past decade – H5N1, H7N9, and H5N6 – all share a couple of important features (see Study: Sequence & Phylogenetic Analysis Of Emerging H9N2 influenza Viruses In China):
- They have all come about through viral reassortment in poultry
- And while their HA and NA genes may differ - they all carry one or more internal genes from the avian H9N2 virus
All of which makes two separate reassortment events in Egypt this year between HPAI H5N8 and LPAI H9N2 very much worthy our attention.
