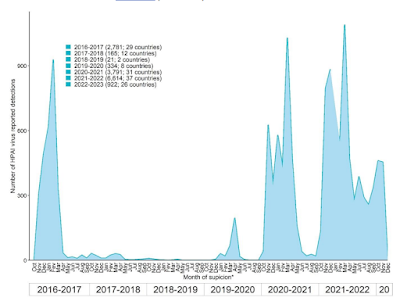
Waves of HPAI H5N1 clade 2.3.4.4b since 2016 - Credit ECDC
#17,198
Unlike the more virulent older clades of avian H5N1, all 4 reported human infections with the H5N1 clade 2.3.4.4b virus that is currently spreading in Europe, North America, Asia, and most recently South America have been mild, or asymptomatic.
While comforting, we've seen numerous reports of severe infections in mammalian wildlife and continued evidence of the virus's evolution, including signs of mammalian adaptations.
At the same time, over the past three months both China and Vietnam have reported isolated, severe human infections (1 fatal) with either H5N1 (China), or H5 (Vietnam), without specifying the clade. Given the location (Asia), and the severity, an older clade of H5 seemed a reasonable assumption.
But today we learn from a WHO Rapid Risk Assessment that both were H5N1 clade 2.3.4.4b. The assessment, dated Dec 21st, only appears to have been posted in the past 24 hours.
These recent severe cases likely explain the spate of risk assessments, and guidance documents, coming from various public health entities the past couple of months (see UK HSA Technical Briefing: Risk Assessment On HPAI H5N1 & Human Infection).
While there is no evidence of widespread infection or human-to-human transmission, this raises the stakes should the virus begin to spread more efficiently.
Excerpts from the WHO announcement follow:
Rapid Risk Assessment
Assessment of risk associated with recent influenza A(H5N1)clade 2.3.4.4b viruses
Background
During 2020, highly pathogenic avian influenza (HPAI) A(H5N1) clade 2.3.4.4b viruses arose from previously circulating A(H5Nx) viruses and spread predominantly via migratory birds to many parts of Africa, Asia and Europe. The epizootic has led to unprecedented numbers of deaths in wild birds and caused outbreaks in domestic poultry. In late 2021, these viruses crossed to North America and subsequently South America in the autumn of 2022. Additionally, there has been an increased spill over to non-avian species including wild terrestrial and marine mammals and, more recently, the detection of an outbreak in a mink farm in Spain. From 2020 to date, six human cases of influenza A(H5N1) belonging to the 2.3.4.4b clade were reported to WHO.
The majority of the influenza A(H5N1) HPAI characterized genetically since 2020 related to these outbreaks are belonging to the 2.3.4.4b clade. This risk assessment focuses on the most recent A(H5N1) viruses belonging to the 2.3.4.4b clade.
Understanding of the virus
Human infections with influenza A(H5N1) 2.3.4.4b viruses
Since the beginning of 2020, detections in humans of influenza A(H5N1) clade 2.3.4.4b viruses have been reported to WHO from the following countries1 : China (one case)[1], Spain (two cases)[2], the United Kingdom of Great Britain and Northern Ireland (one case)[3], the United States of America (USA) (one case)[4], and Viet Nam (one case)[1].
All four human cases reported in Europe and North America were asymptomatic or mild, with only fatigue reported for the case detected in the USA. The case detected in China resulted in a fatality while the case in Viet Nam had severe symptoms but recovered. All human cases had exposure to infected poultry either through participation in response activities to poultry outbreaks or direct exposure to infected poultry in backyard holdings or live bird markets.
Virus sequences from these human cases, where available, did not show markers for mammalian adaptation nor for resistance to neuraminidase inhibitors (such as oseltamivir) or endonuclease inhibitors (such as baloxavir).
Based on the available information for A(H5), although based on limited seroprevalence information available on other A(H5) virus subtypes and clades, human population immunity against the A(H5) clade 2.3.4.4b virus haemagglutinin is expected to be minimal.
Infections in animals
Avian influenza A(H5N1) viruses, especially those in clade 2.3.4.4b, continue to diversify genetically and spread geographically. From 2021 to 2022, Europe and North America have observed their largest and most extended epidemic of avian influenza with unusual persistence of the virus in wild bird populations. A broader range of wild bird species continue to be infected globally which has significant ecological consequences and has caused mass die offs in some species.
Additionally, continuous infection in wild and migratory birds has led to multiple separate incursions in domestic species. These circumstances have led to increased opportunities to generate multiple genotypes with varied clinical signs. Some of the recent viruses have caused severe infections with neurological signs in mammals.[5]
There have been limited reports of transmission between mammals despite the increase in mammalian infections. Affected mammals include badger, black bear, bobcat, coyote, dolphin, ferret, fisher cat, fox, lynx, mink (mink-tomink in Spanish farm), opossum, otter, pig, polecat, porpoise, raccoon, raccoon dogs, seal (seal-to-seal in USA) and skunk.
Regular monitoring and screening of viral sequences found few sequences with markers of mammalian adaptation. These mutations likely occurred after transmission to the mammalian host and do not seem to transmit onwards. Continuous monitoring is warranted to understand if these changes continue to occur or accumulate over time. Available A(H5N1) clade 2.3.4.4b virus sequences from avian and mammalian hosts indicate that markers associated with reduced susceptibility to neuraminidase or endonuclease inhibitors are rare.
From published animal transmission studies, transmission between ferrets did not occur, however some genotypes resulted in severe disease in infected ferrets.[6, 7]
Candidate vaccine viruses
The WHO Global Influenza Surveillance and Response System (GISRS), in collaboration with animal health and veterinary sector colleagues, regularly evaluate candidate vaccine viruses. Clade 2.3.4.4b A(H5) candidate vaccine viruses (CVV) have been developed. This includes a A(H5N8) clade 2.3.4.4b CVV made from A/Astrakhan/3212/2020 as well as a newly recommended A(H5N1) A/chicken/Ghana/AVL-76321VIR7050- 39/2021-like virus which is under development. The HA of A/Astrakhan/3212/2020 is closely related to the circulating strains
Summary of the assessment of current risk to humans posed by influenza A(H5N1) clade 2.3.4.4b viruses
Despite the high number of poultry outbreaks and likely human exposures to the virus at the human-animal environment interface since 2020, only six A(H5N1) clade 2.3.4.4b virus detections in samples from people directly exposed to infected poultry have been reported. In the four human cases from Europe and North America, the individuals had no symptoms or only mild clinical signs, however the two cases from Asia had severe and fatal outcome. The use of antivirals as part of the treatment of the severe and fatal cases is unknown.
Recently, there was an increase in reports of spill over from wild birds to some mammalian species in different countries in Europe and North America. This is likely a result of high prevalence of the virus in avian populations in these regions. There is still limited evidence for mutations associated with adaptation to mammals and humans even when transmission in mammals has been reported. At this juncture, the risk of infection for humans remains low and no sustained human-to-human transmission has been reported.
A week ago, in the ECDC/EFSA Avian influenza overview September – December 2022 we learned that 8 new genotypes have appeared in Europe over the past 3 months, while in November we saw several new genotypes described in North America.
These new genotypes can vary greatly in their ability to infect avian and non-avian hosts, and in their virulence.
It is a pretty safe assumption that in Asia, South America - and in many other parts of the world - additional genotypes are emerging, mostly outside of our view. Most will provide no special advantage to the virus, and some may actually be detrimental to its survival.
While the future evolution of these avian viruses is unknowable - and there may even be a `species barrier' that prevents an H5 virus from sparking a human pandemic (see Are Influenza Pandemic Viruses Members Of An Exclusive Club?) - our recent luck in these matters hasn't been particularly reassuring.But the potential exists for the emergence of a biologically fit, and more dangerous virus.
Which is why we need to be preparing now for the next global health crisis, because another pandemic is inevitable.
We just don't know when.