#17,318
Last October Spain reported a large outbreak of avian flu at a mink farm (Spain: Avian H5N1 Spillover Into Farmed Mink), which was followed up in January by a Eurosurveillance report that found evidence of a rare mammalian adaptation - (T271A), which `enhances the polymerase activity of influenza A viruses in mammalian host cells and mice' - in the virus.
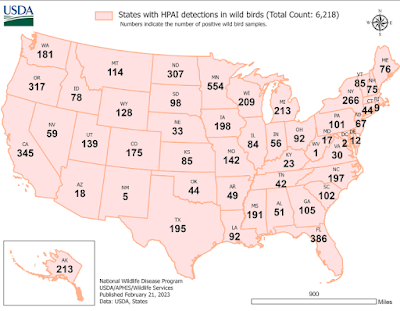
Elsewhere, over the past 18 months we've seen a growing number of spillovers of H5N1 into both terrestrial and aquatic mammals, sometimes producing large die offs (see Peru Reports At Least 585 Sea Lions killed by Avian Flu).
While still primarily adapted to birds, the concern is that over time, the virus could better adapt to mammals (including humans), increasing its pandemic threat. How likely that is to happen is unknown.
Farmed mink are members of the Mustelidae family of carnivorous mammals, which also includes otters, badgers, weasel, martens, ferrets, and wolverines, many of which are susceptible to both influenza, and more recently SARS-CoV-2.
We reviewed this history most recently in That Touch of Mink Flu (2023 Edition), with reports going back nearly 40 years.
In late 2020 Danish authorities were alarmed when they discovered several mutated strains of COVID-19 had arisen in farmed mink and had transmitted to humans (see Denmark Orders Culling Of All Mink Following Discovery Of Mutated Coronavirus).
This emergency was relatively short-lived, as the Alpha variant emerged in Europe in late 2020 and quickly supplanted these mink-variants. But it did illustrate the problem; carriage of SARS-CoV-2 by other host species can produce new variants, which can jump back into humans.
While Denmark temporarily banned the commercial raising of mink following that incident, that ban expired on January 1st, 2023, although the government expects `significantly reduced' mink production going forward.
Today Denmark's SSI, along with the Danish Veterinary Consortium, has published a 19-page risk assessment on the risks of HPAI H5N1 in farmed mink. While it is published in Danish, I've (machine) translated some excerpts below.
While generally reassuring, you'll note that their confidence level for a number of their conclusions is low.
First the brief announcement from the SSI:
Low to moderate risk of infection with bird flu from mink to humans
In connection with a new risk assessment on the likelihood of Danish mink becoming infected with bird flu, the Statens Serum Institute has also assessed the risk of a potential infection among mink being transferred to humans.
Last edited on February 28, 2023
SSI assesses that the risk of infection to humans from mink is low to moderate in the event of exposure to infected mink. This assessment is in line with assessments from ECDC and WHO.
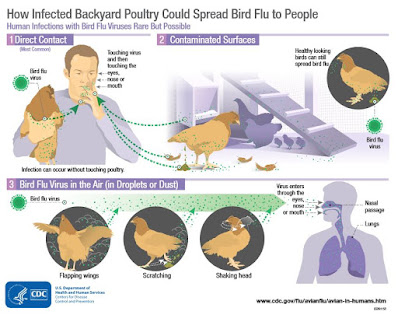
The risk of humans becoming infected is greatest if the virus spreads between mink. Early detection of viruses in mink and humans is essential to reduce the overall health risk.
In addition, the mink breeders can protect themselves and limit the risk of infection from mink with bird flu by using masks, washing hands and changing clothes before entering and leaving the farms, as they must in advance to avoid infection with covid- 19.
Read the risk assessment from the Danish Veterinary Consortium
(Translated Excerpts)
Summary
An infection of mink with highly pathogenic avian influenza (HPAI) subtype H5N1 has been confirmed, with subsequent signs of infection spreading among the mink on a mink farm in Spain in October 2022. HPAI is notifiable in Denmark if the disease is suspected in all animals, thus also in mink. In this connection, the Danish Veterinary and Food Administration must decide on the further handling of HPAI in relation to the recently resumed mink production in Denmark. For this purpose, the Danish Veterinary and Food Administration has requested the Danish Veterinary Consortium's (DK-VET's) assessment of a number of issues arising from the Spanish outbreak. DK-VET's experts have reviewed the available evidence and carried out a qualitative risk assessment with contributions from the human area at Statens Serum Institut (SSI).
There is limited knowledge about the occurrence and course of HPAI virus infections in mink. DK-VET assesses that the probability of introduction of HPAI in Danish mink primarily depends on the occurrence of HPAI in the wild birds in the area in question and will therefore vary over the year. The occurrence of HPAI in wild birds is usually highest in the winter, although in recent years HPAI has been detected in wild birds all year round. Furthermore, the risk for the individual farm will depend on the layout, size and location. For farms located close to areas where there are many wild birds and which consist of open halls where there is access for wild birds, the risk of infection of Danish mink with HPAI is generally assessed to be unlikely (1-10 % ) (cf. EFSA criteria) during periods of high HPAI activity in wild birds. For farms with a high level of infection protection, where there is limited access for wild birds, and for all farms in periods of the year with low HPAI activity in wild birds, the risk is assessed to be extremely unlikely (0.001-0.1%) (cf. EFSA criteria).
These estimates represent respectively the highest and lowest probability for the individual farm, but there will be farms where the probability of infection lies between the two estimates. The highest probability is estimated to be in the period October-November. Import of mink from abroad is considered to pose a lower risk of introduction of HPAI to Danish mink compared to the probability of mink being infected with HPAI from birds in Denmark.
The greatest risk of infection of mink with the HPAI virus is estimated to be through direct or indirect contact with infected wild birds, including seagulls. The hygienic infection prevention measures introduced to limit the risk of SARS-CoV-2 infection of mink from humans and the environment will also contribute to reducing the risk of introduction of the HPAI virus in mink and may contribute to reducing the risk of infection by humans in the event that mink become infected with the HPAI virus. However, this is on the condition that the infection prevention measures are all implemented and that there is full compliance. Specific measures in herds, which are established with a view to reducing the probability of wild birds and other wild animals coming into direct and indirect contact with the minks, will help to reduce the probability of introduction of the HPAI virus.
It is assessed that the risk of infection to humans from mink is low to moderate (cf. ECDC criteria) in the event that there is exposure to HPAI-infected mink. The risk of people becoming infected is greatest if the virus spreads between mink on the farm, as it is theoretically possible that virus variants with an increased ability to infect and between people are developed and thus an increased risk of community infection. Early detection of HPAI virus in mink and humans is therefore essential to reduce the overall health risk.
(SNIP)
Conclusion and perspective
The risk of HPAI virus infection in humans from mink can be divided into three different risks: the risk of mink becoming infected with the HPAI virus, the risk of the mink subsequently infecting humans with a non-adapted virus and the risk of virus variants developing in mink that have increased risk of infection to and between people (adapted viruses).
Infection of mink with HPAI virus
The risk of introduction of HPAI in Danish mink depends on the occurrence of HPAI in the wild birds in the area in question and will therefore vary over the year. Furthermore, the risk for the individual farm will depend on a number of herd-specific factors. For farms located close to areas where there are many wild birds and which have a low level of infection protection, the probability of infection is assessed to be unlikely (1-10%) in periods of high HPAI activity in wild birds. For farms with a high level of external infection protection and for all farms in periods of low HPAIV activity in wild birds, the probability is assessed to be extremely unlikely (0.001-0.1%). The highest risk is assessed to be in the period October November when there is both high HPAI activity and many animals on the farms. Import of mink from abroad and introduction from other mink farms in Denmark is considered to pose a lower risk of infection of Danish mink compared to the risk of infection via wild birds in Denmark.
The greatest risk of infection of mink with the HPAI virus is estimated to be through direct or indirect contact with infected wild birds, including seagulls. Specific measures in herds, which are established with a view to reducing the risk of wild birds coming into direct and indirect contact with the minks, will help to reduce the risk of introduction of the HPAI virus.
A number of the measures introduced to limit the risk of SARS-CoV-2 infection of mink will also reduce the risk of introduction of the HPAI virus as well as contribute to reducing the risk of infection of humans in the event that mink are infected with HPAI virus. Just as the passive surveillance for SARS-CoV-2 with the obligation to report clinical signs of SARS-CoV-2 simultaneously can be used as a possible indicator of infection with HPAI, in that the clinical signs overlap to a large extent. However, this requires that samples taken from clinically ill mink are also tested for HPAI. Similarly, mink workers who are currently recommended SARS-CoV-2 tests for respiratory symptoms will also be able to be offered tests for influenza.
Transmission to humans from mink
The risk of people becoming infected is greatest if the virus spreads between mink on the farm, as people are then exposed for a longer period of time.
The overall risk of infection to humans exposed to HPAI-infected mink is assessed to be low to moderate. It is also estimated that some of the infection protection measures introduced in relation to SARS CoV-2 will also have an effect on HPAI infection.
Development of virus variants in mink that have an increased risk of infection to and between humans (adapted viruses)
There is a theoretical risk that by passage in mink over a longer period of time variants may be developed that are better adapted to mammals and therefore perhaps also to humans. Studies of avian influenza A virus infection in other animals, including wild mammals and ferrets, have shown that adaptive mutations occur quickly both at first exposure and when the virus passes from animal to animal (20) . In a situation where mink have already been infected with HPAI and the virus spreads between mink, it is considered to be very likely (cf. EFSA criteria) that variants of HPAI can develop that are adapted to mammals and thereby also potentially adapted to people. The certainty of this assessment is low. The consequence in the worst case will be that a variant of the virus arises that can be transmitted from person to person, and thus trigger widespread social contagion.
Early recognition of the HPAI virus in mink and humans is therefore essential to reduce the overall health risk.