Just over 500 Cases in 15 Years in the United States
#18,129
Over the past dozen years we've seen more than 500 swine variant cases (H1N1v, H1N2v, H3N2v) reported by the CDC (see chart above), the vast majority (88%) of which were H3N2v. Prior to 2016, the H1N2v subtype was only rarely reported (n=7), but since then it has been appearing with increasing frequency (n=38).
- Last winter, the UKHSA Announced the 1st H1N2v (Swine Variant) Infection In the UK, and in In January the ECDC's journal Eurosurveillance published a Rapid Comms on this event, which described 1 laboratory confirmed - and two probable - infections that were picked up by the UK's epidemiological investigation.
- Over the past 3 years Taiwan has reported 3 human H1N2v infections (see Taiwan CDC Reports 3rd H1N2v Case)
- In 2021 alone, Canada reported 4 cases and we've seen scattered cases reported from Brazil, and France.
Swine variant influenza infections are indistinguishable from seasonal flu without highly specialized testing, which few countries tend to do. Even here in the United States it is assumed we only detect a small percentage of cases (see CID Journal: Estimates Of Human Infection From H3N2v (Jul 2011-Apr 2012)).
H1N2 variant [A/California/62/2018] Jul 2019 5.8 5.7 Moderate
H3N2 variant [A/Ohio/13/2017] Jul 2019 6.6 5.8 Moderate
H3N2 variant [A/Indiana/08/2011] Dec 2012 6.0 4.5 Moderate
Highlights
- Five quadruple reassortant swH1N2 viruses were identified which have close genetic relationship with current human virus.
- The swH1N2 viruses replicate effectively in human, canine, and swine cells and mice.
- A/swine/ Shandong/198/2020 replicated efficiently in the respiratory tract of pigs and spread effectively.
- The swH1N2 viruses pose zoonotic potential, highlighting the need for strengthened surveillance of swH1N2 viruses.
Potential pandemic risk of circulating swine H1N2 influenza viruses
Valerie Le Sage, Nicole C. Rockey, Andrea J. French, Ryan McBride, Kevin R. McCarthy,Lora H. Rigatti, Meredith J. Shephard, Jennifer E. Jones, Sydney G. Walter, Joshua D. Doyle, Lingqing Xu, Dominique J. Barbeau, Shengyang Wang, Sheila A. Frizzell, Michael M. Myerburg, James C. Paulson, Anita K. McElroy, Tavis K. Anderson, Amy L. Vincent Baker & Seema S. LakdawalaNature Communications 15, Article number: 5025 (2024)
Influenza A viruses in swine have considerable genetic diversity and continue to pose a pandemic threat to humans due to a potential lack of population level immunity. Here we describe a pipeline to characterize and triage influenza viruses for their pandemic risk and examine the pandemic potential of two widespread swine origin viruses.
Our analysis reveals that a panel of human sera collected from healthy adults in 2020 has no cross-reactive neutralizing antibodies against a α-H1 clade strain (α-swH1N2) but do against a γ-H1 clade strain. The α-swH1N2 virus replicates efficiently in human airway cultures and exhibits phenotypic signatures similar to the human H1N1 pandemic strain from 2009 (H1N1pdm09).
Furthermore, α-swH1N2 is capable of efficient airborne transmission to both naïve ferrets and ferrets with prior seasonal influenza immunity. Ferrets with H1N1pdm09 pre-existing immunity show reduced α-swH1N2 viral shedding and less severe disease signs.
Despite this, H1N1pdm09-immune ferrets that became infected via the air can still onward transmit α-swH1N2 with an efficiency of 50%. These results indicate that this α-swH1N2 strain has a higher pandemic potential, but a moderate level of impact since there is reduced replication fitness and pathology in animals with prior immunity.
(SNIP)
Discussion
Identification of emerging respiratory viruses with pandemic potential is critical for enacting preparedness measures to mitigate their impact. Swine viruses are particularly concerning, given their agricultural importance that places them within close physical proximity to humans and the wide diversity of swine influenza strains48. Current risk assessment of pandemic threats is done through the WHO and CDC risk assessment tools49,50, which use subject-area expert opinion to assign weighted scores for various categories and limited experimental data derived from multiple different in vitro and in vivo sources.
In this study, we present a streamlined, adaptable strategy to experimentally triage influenza viruses that reduces the need for complete virus characterization since certain criteria must be met before proceeding to the next box in the decision tree. This pipeline represents a breathable framework that can and will be updated as additional data from characterization studies are conducted.
Using our decision tree, we analyzed representative circulating swine H1 strains from the alpha and gamma genetic clades that have a wide geographic distribution, are frequently detected in swine populations in the United States (Fig. 1C), and have exhibited sporadic human spillover events51. Previous representatives of the α-swH1N2 clade were shown to have antigenic distance from human vaccine strains, reduced recognition by human sera from two different cohorts6, and transmitted efficiently from infected pigs to naive recipient ferrets52.
While highly efficient at controlling antigenically similar influenza viruses, antibodies directed towards HA become less effective over each subsequent flu season as surface glycoproteins rapidly mutate through antigenic drift. No cross-neutralizing antibodies were detected against α-swH1N2 in H1N1pdm09- or H3N2-imm ferrets (Supplementary Table 2), suggesting that an initial infection with human seasonal viruses does not produce antibodies that cross-neutralize, and this was consistent with our human serum data (Fig. 3). Interestingly, human sera across all birth years tested had variable levels of anti-N2 antibodies (Supplementary Fig. 5), which may suggest that this NA-based immunity could provide some level of protection in a subset of the population53,54,55,56.
Our prior work previously determined that prior immunity can influence the susceptibility to heterosubtypic viruses in a mechanism not mediated by neutralizing antibodies40. Thus, prior immunity from divergent strains can impact susceptibility of viruses through the air. We found that α-swH1N2 transmitted efficiently through the air to ferrets regardless of immune status, but the severity of disease after experimental infection with α-swH1N2 was lower in animals with prior immunity. A similar phenomenon may explain the lower-than-expected morbidity and mortality of the 2009 pandemic in humans57.
Protection against emerging influenza virus strains in hosts without neutralizing antibodies can be conferred from CD8+ T cells, which recognize conserved internal influenza virus proteins. Although prior adaptive immunity may not prevent influenza virus infection, CD8+ T cells that display cross-reactivity against different subtypes of influenza virus have been linked to more efficient clearance of virus and faster recovery from illness58,59,60. Indeed, prior immunity to human seasonal viruses was not protective against α-swH1N2 airborne infection (Fig.5B, C).
However, the lack of disease severity in immune animals may also provide an opportunity for this virus to spread undetected and gain a foothold in the population, creating a pandemic risk.
Taken together, our data demonstrate that this α-swH1N2 virus strain poses a higher pandemic risk than γ-swH1N1 that warrants continued surveillance efforts to capture zoonotic events and an increased campaign to vaccinate swine against this H1 clade to reduce the amount of virus in source populations.
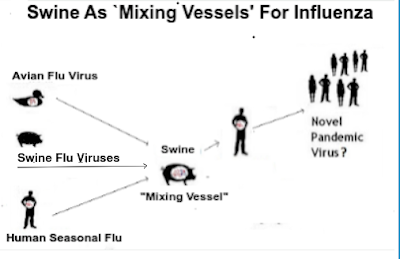
While our attentions this summer are understandably focused on H5N1 in cattle, it is far from the only viral threat we face. We are also entering in to what is traditionally `swine-variant season' - summer and fall - when we usually see the the most spillovers into humans.
These tend to occur at agricultural exhibits at county and state fairs, where large numbers of people have contact with livestock (see EID Journal: Shortening Duration of Swine Exhibitions to Reduce Risk for Zoonotic Transmission of Influenza A Virus).
While most fairs have requirements the screening and barring of `sick pigs' from exhibition, over the years we've learned that many pigs may be infected asymptomatically (see EID Journal: Flu In Healthy-Looking Pigs and Transmission Of Swine H3N2 To Humans At Agricultural Exhibits - Michigan & Ohio 2016).
Although the public health risks are considered low, the CDC advises those who are at higher risk of serious flu complications (including children under 5, adults over 65, pregnant women, and those with certain chronic medical conditions), to avoid pigs and the swine barn altogether.
There are understandable concerns now that H5N1 could enter the mix of viruses spreading among pigs (see EID Journal: Divergent Pathogenesis and Transmission of Highly Pathogenic Avian Influenza A(H5N1) in Swine).
While we don't know exactly how that would turn out, it could provide the HPAI virus with the building blocks to reassort into a more formidable foe.