Earlier this week, in Eurosurveillance: Cryptic SARS-CoV-2 Lineage Identified on Two Mink Farms In Poland, we looked at the detection of two closely related COVID variants that turned up - 3 months apart - at two mink farms in Poland.
Both closely matched a (pre-Omicron) variant (B.1.1.307) not seen in humans in nearly two years. At the same time both showed clear signs of continued evolution (at least 40 nt changes) since its last appearance in humans.
This suggests that an older SARS-CoV-2 variant has been circulating unnoticed in another (non-human) host in Northern Europe for nearly 2 years, and that it has spilled back (twice) into farmed mink. The identity of this host isn't known, although wild mink, cats, dogs, rodents, or other peridomestic mammals are all possibilities.
Luckily, this returning variant has not been seen (yet) in humans.
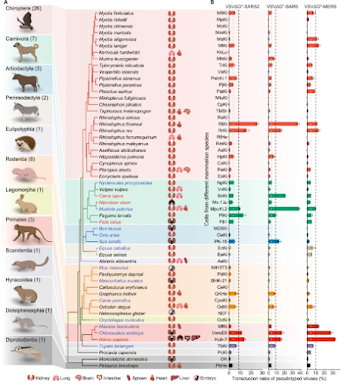
While overwhelmingly a `humanized' pathogen, SARS-CoV-2 has also found a plethora of other suitable hosts, including mink, deer, dogs, cats, and even rodents (see Nature: Comparative Susceptibility of SARS-CoV-2, SARS-CoV, and MERS-CoV Across Mammals).
Companion animals (cats and dogs) are susceptible to infection, and they are of particular interest because they are uniquely positioned to be exposed to infected humans, and they are also likely to serve as a conduit both to - and from - other wildlife species.
Ten days ago, in EID Journal: Human-to-Animal Transmission of SARS-CoV-2, South Korea, 2021, we saw a study that found the incidence of COVID infection of household pets may be much higher than previously suspected. The authors wrote:
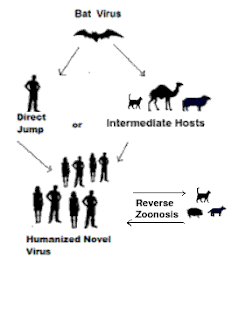
This study reveals SARS-CoV-2 positivity rates of 24.0% for dogs and 35.6% for cats in South Korea, higher than rates previously reported from studies of dogs and cats. Although the animals in our study were already known to have been exposed to SARS-CoV-2 because their owners were confirmed to have COVID-19, the rate of positivity is high compared with rates determined in previous studies of animals with SARS-CoV-2–positive owners (8,9). This finding emphasizes the value and necessity of managing infectious diseases in companion animals as well as in humans because the risk for reverse zoonoses increases when companion animals are in prolonged and close contact with their owners.
Unlike the above study, which tested companion animals in household with confirmed (and current) COVID cases, the following report is on a seroprevalence study in Spain that tested a wide range of dogs and cats (including strays), looking for SARS-COV-2 antibodies.
Interestingly, all of the stray animals that showed antibodies (n=6) were cats. First the link, abstract, and some excerpts from the report. I'll return with a bit more after the break.
Research
SARS-CoV-2 Seroprevalence Studies in Pets, Spain
Sandra Barroso-Arévalo, Lidia Sánchez-Morales, Jose A. Barasona, Lucas Domínguez, and José M. Sánchez-Vizcaíno
Abstract
SARS-CoV-2 can infect domestic animals such as cats and dogs. The zoonotic origin of the disease requires surveillance on animals. Seroprevalence studies are useful tools for detecting previous exposure because the short period of virus shedding in animals makes detection of the virus difficult.
We report on an extensive serosurvey on pets in Spain that covered 23 months. We included animals with exposure to SARS-CoV-2–infected persons, random animals, and stray animals in the study. We also evaluated epidemiologic variables such as human accumulated incidence and spatial location. We detected neutralizing antibodies in 3.59% of animals and showed a correlation between COVID-19 incidence in humans and positivity to antibody detection in pets.
This study shows that more pets were infected with SARS-CoV-2 than in previous reports based on molecular research, and the findings highlight the need to establish preventive measures to avoid reverse zoonosis events.
(SNIP)
Previous studies on pets in Spain have demonstrated a low prevalence of positive animals by PCR (11,20). However, as we have demonstrated, more animals have been exposed to the virus. In all those cases, we can confirm that the exposure resulted in an active infection because the animals were able to develop an effective immune response based on neutralizing antibodies. We suspect that SARS-CoV-2 infection in pets is anecdotic because in none of the positive cases we described did the owners detect severe symptoms in the animals.
Although some animals had antibodies and were experiencing clinical signs at the time of sampling (30.3%), such as sneezes, dyspnea, nasal discharge, coughs, vomiting, or depression, the relationship between those signs and the SARS-CoV-2 infection is not clear enough. Antibodies remain undetectable in serum until 8–10 days postinfection (19), leading to a delay between a positive result to antibody detection and the infection. In addition, a high percentage of the animals were sampled during their attendance at the veterinary clinic, and the symptoms reported as the reason for the visit might be unrelated to SARS-CoV-2 infection; comorbidities may cause a biased result.
We confirmed that stray animals had neutralizing antibodies, as do domestic animals in contact with SARS-CoV-2–infected persons. Those results are in line with those from other studies that confirmed the presence of neutralizing antibodies in stray animals (31–33). However, the seroprevalence in this group of animals was very low; the domestic animals represented 4.05% of the animals with neutralizing antibodies, compared with 1.69% in the case of stray animals. Those results make sense because domestic animals are more likely to be in contact with infected persons and share potentially contaminated spaces than stray animals. In those cases, the exposure to the virus may be related to the times that humans fed the stray colonies and to the presence of infectious excretions in the areas frequented by stray cats and dogs. Another potential route of transmission is animal-to-animal transmission, which has been demonstrated in the case of stray cats (9). These results suggest that virus circulation in stray populations is low, although special care should be taken in practices that may pose a risk, such as the feeding of stray animals.
In conclusion, this study demonstrated higher rates of human-to-pet SARS-CoV-2 transmission than those found by direct molecular detection. As expected, the seroprevalence of the disease was higher in animals with previous exposure to infected persons, whereas the lower risk of infection in stray animals is likely caused by a low rate of exposure. In addition, the epidemiology of the disease in the human population seems to influence the seroprevalence of the infection in cats and dogs, which highlights the importance of performing active surveillance in susceptible species.
Dr. Barroso-Arévalo is a postdoctoral researcher at the SUAT-VISAVET team of the Complutense University of Madrid. Her research interests include SARS-CoV-2 and African swine fever viruses, general virus pathogenesis, immunology, and vaccine development.
It is worth noting that not all humans (and the same likely holds true for other hosts) develop a strong and long-lasting antibody response to a SARS-CoV-2 infection, meaning the actual number of infected pets is likely higher.
Last November, a preprint (see Wildlife Exposure to SARS-CoV-2 Across a Human Use Gradient) found evidence of SARS-CoV-2 infection across a wide variety of small peridomestic mammals (e.g. possums, skunks, squirrels, etc.) in Virginia, while another study found evidence of SARS-CoV-2 Exposure in Norway Rats in New York City.
Our knowledge of what SARS-COV-2 is doing in other species remains limited, with companion animals, captive animals, and farmed animals getting the most attention (see USDA data below).
While we appear to be tantalizingly close to moving into a post-pandemic phase with COVID, the emergence of a new, more dangerous variant from a non-human reservoir could prove to be a serious setback for the world.
Which is why we need to be prepared for more COVID-related surprises in the months, and years, to come.