#17,519
It has been 9 days since the first media reports of unusual cat illnesses and deaths began to come out of Poland (see Media Reports Of Unusual Cat Deaths In Poland), and understandably, the spillover of avian flu to dozens (perhaps scores) of companion animals has raised concerns globally.
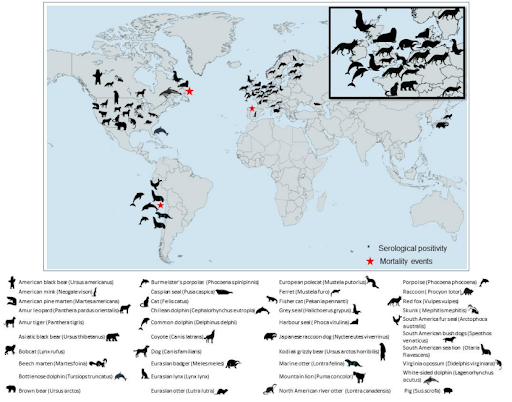
As we've seen with other mammalian infection with this new H5N1 clade (2.3.4.4b), it is often fatal, and frequently involves profound neurological symptoms (see Emerging Microbes & Inf.: Neurotropic HPAI H5N1 Viruses with Mammalian Adaptive Mutations in Free-living Mesocarnivores in Canada).
While no human cases have been reported, surveillance and testing for novel flu viruses is limited, and it is possible some cases have been missed.
Given what we know - and more importantly, what we don't know - the ECDC's risk assessment below is understandably incomplete. They are, however, recommending pet owners, and vets, take precautions.
1. Avian influenza in domestic cats - Poland -2023
Overview: Update On 28 June 2023, the Polish Chief Veterinary Officer (CVO) issued a press release notifying about a total of 16 cat samples positive for influenza A(H5N1) in the country. Positive samples originate from many cities in Poland (Gdańsk, Gdynia, Pruszcz Gdański, Lublin, Bydgoszcz, Poznań and Warsaw).
The World Organisation for Animal Health (WOAH) issued a statement on the Polish cat outbreak mentioning that the severe and rapid course of the infection is consistent with reports of A(H5N1) infection in Felidae, and noting that several cases of infection in cats with Influenza A(H5N1) have been reported from Europe and North America in the context of the ongoing panzootic. WOAH stresses the need for more investigations and states that since the cases include both stray and pet cats, exposure to sick wild birds is not considered a likely transmission mode.
In addition, the wide geographical distribution of cases suggests that the primary mode of spread in these cases is not cat-to-cat transmission but rather some other kind of common source. The statement also mentions the need to isolate any suspected cases from other pets due to potential shedding from the gastrointestinal tract, and the need for appropriate personal protective equipment (PPE) for the handlers of such animals.
Media sources from Poland cite the director of the National Veterinary Institute in Puławy stating that "they have detected two mutations that indicate that the A(H5N1) virus is evolving to multiply more easily in mammals".
Summary
Since 23 June 2023, media sources referred to several deaths of domestic cats (at least 70) in Poland for which investigations are ongoing. On June 26, the Chief Veterinary Officer of Poland announced in a press release that 11 samples were tested at the National Veterinary Institute in Puławy, of which nine were positive for A(H5N1) influenza virus. The positive samples come from the cities of Poznań, Tricity and Lublin. Sequencing is ongoing. Preliminary studies have ruled out connection to the avian influenza outbreaks in seagulls, that Poland has registered in recent weeks.
No source of infection has been identified yet. An intersectoral meeting between animal and human public health services took place on 26 June. The CVO press release included advice to the public to prevent contact of pet cats with other animals, including keeping them inside homes and avoid contact with footwear used outside, enhanced hand hygiene for all pet cat owners is also advised.
ECDC assessment: Several uncertainties currently exist as regards the source of infection, the potential of feline-to-feline and felineto-human transmission of the particular A(H5N1) influenza virus strain, as well as regards to the severity of the disease. No human cases have been reported related to this event, however, there are too many uncertainties at this stage to properly assess the risk to the general public.
ECDC will review its assessment as soon as more information becomes available.
Actions: ECDC is monitoring this event and has contacted Polish public health authorities and EFSA for further investigation. Last time this event was included in the CDTR: 29 June 2023
So far we've only seen reports of mass cat infections from Poland, but that could easily change. Hopefully neighboring countries are ramping up surveillance, although that likely isn't an option in Ukraine right now.
For more on the difficulties of detecting novel flu cases in the community, you may wish to revisit UK Novel Flu Surveillance: Quantifying TTD.
On Monday of this week the ECDC released timely Guidance: Enhanced Surveillance of Severe Avian Influenza Virus Infections in Hospital Settings, and last week the CDC presented a COCA Call for clinicians on What Providers Need to Know about Zoonotic Influenza.
Stay tuned.